The novel coronavirus, Covid-19, has rapidly spread around the world, overwhelming healthcare systems, shutting down countries and causing widespread grocery shortages. Understandably, this has led to fear and misinformation about the Covid-19 virus and potential treatment options.
The good news is that many biopharmaceutical companies are working towards new treatments, cures and preventive vaccines for Covid-19. There is hope that our industry will be able to tackle this virus and solve one of the biggest challenges facing the world today.
The Industry is Ready to Take on This Challenge
Companies usually need a decade or longer to develop a new drug. However, after SARS, MERS and Ebola, the biopharmaceutical industry has been working quietly behind the scenes to improve preparedness for future pandemics. Organizations such as the Bill and Melinda Gates Foundation and the Coalition for Epidemic Preparedness Innovations offer funding and facilitate collaborations between industry stakeholders.
The positive effects of these measures is evident in the current pandemic. Scientists have sequenced the novel coronavirus responsible for this outbreak and companies are now testing a number of therapeutic and vaccine candidates only months after Covid-19 was first identified.
Collaborations
One of the most positive things to come out of this pandemic is widespread international and public-privation collaboration across the biopharmaceutical industry. There are now too many partnerships to list here, but as a few examples:
- GSK is collaborating on two separate Covid-19 vaccines with the University of Queensland in Australia and Chinese pharmaceutical company, Clover.
- Moderna is collaborating with the National Institute of Allergy and Infectious Diseases (NIAID) on their mRNA vaccine.
- AbbVie is working with the WHO, CDC and National Institutes of Health to repurpose their antiviral combination lopinavir/ritonavir.
- The U.S Biomedical Advanced Research and Development Authority (BARDA) is working with pharma companies, including Johnson & Johnson and Sanofi.
Drugs to Fight Covid-19
Scientists are using several different approaches against Covid-19, including repurposing drugs, searching for new therapeutics and developing novel vaccines.
Antivirals
Antiviral drugs directly target disease-causing viruses. One approach that may successfully combat the current coronavirus pandemic is to repurpose existing antiviral drugs, such as Gilead’s remdesivir, developed to treat Ebola, and AbbVie’s Kaletra (lopinavir/ritonavir), approved to treat HIV. Any drugs that have already been approved for use in humans are likely to advance through trials more quickly than novel drug candidates. Other companies such as Pfizer have discovered new antiviral molecules that can control the Covid-19 virus in vitro.
Antibodies
Monoclonal antibody therapies are commonly used to treat cancer, and a number of different antibody drug candidates are in development to treat various viral diseases, including the new coronavirus. Regeneron and Sanofi will test whether Kavzara (sarilumab), an antibody therapy for rheumatoid arthritis, can prevent lung inflammation in Covid-19 patients. Regeneron, Eli Lilly and AbCellera are also working to find novel antibodies to treat Covid-19 patients. U.S company Vir has partnered with Chinese company WuXi Biologics to identify antibodies in SARS patients might work against Covid-19.
Vaccines
Vaccines could help protect large populations against the novel coronavirus. Moderna has developed an mRNA vaccine that stimulates an immune response against the Covid-19 virus. Inovio Pharmaceuticals and Sanofi are also developing Covid-19 vaccines.
Others
Other classes of drugs may also be effective against the Covid-19 virus, including Arava (leflunomide), a disease-modifying anti-rheumatic drug, anti-fibrotic drug, Pirfenidone (esbriet), and the malaria drug, chloroquine.
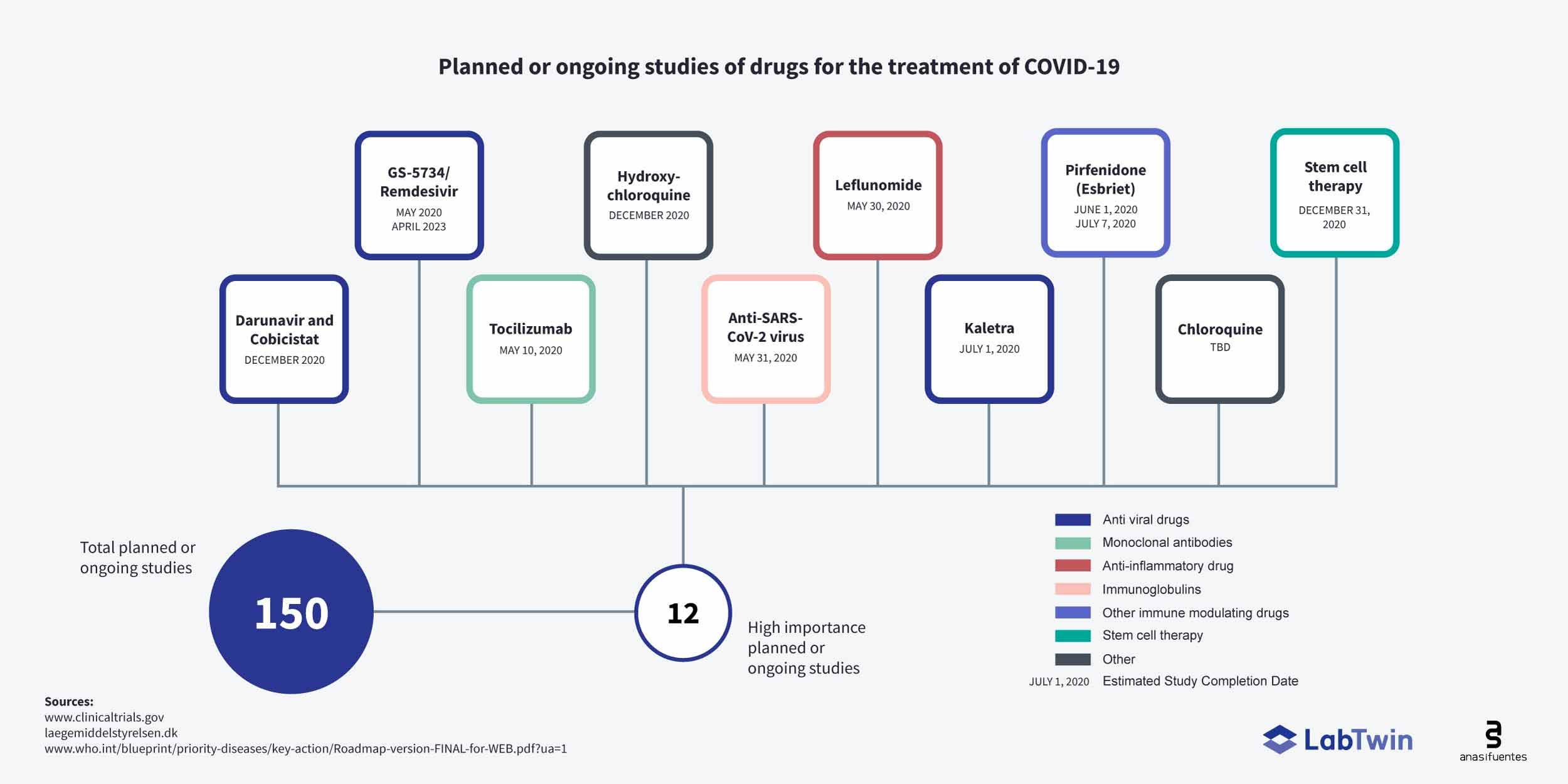
Current and Planned Clinical Trials
There are now hundreds of ongoing or planned clinical trials to test drugs and vaccines in Covid-19 patients. Clinical trials for remdesivir are underway in China and the U.S, and new trials are planned in other countries battling Covid-19 outbreaks. A trial of the HIV drug combination, Kaletra, is planned to start in July, 2020.
A clinical trial of the rheumatoid arthritis drug, Arava (leflunomide) is planned for May this year. Pirfenodone (esbriet), approved for use in idiopathic pulmonary fibrosis, will also be trialed in Covid-19 patients in June-July, 2020.
Moderna’s mRNA vaccine is now underway. Inovio may start testing their DNA vaccine candidate in April, 2020.
Conclusion
The biopharmaceutical industry is working hard to find new therapeutics and preventive measures against the Covid-19 pandemic. Ongoing and planned clinical trials will test several different classes of drugs, and drug combinations, in the hope of lessening the negative impact of this new disease. The lessons we learn from dealing with this pandemic will hopefully help us manage any future outbreaks.