GMP sample preparation involves many complex steps including weighing, dissolving, diluting, incubating etc. Technicians must perform each step with precision in a clean or sterile environment. The specific techniques employed depend on both the type of sample and how it will be used. This variation makes it difficult to automate sample preparation across a manufacturing facility. Therefore, a lot of sample preparation is still done manually.
Many facilities also use manual data collection or hybrid systems with both paper and electronic tools such as a Manufacturing Execution System (MES) or Laboratory Information Management System (LIMS). MES and LIMS are often located on computers away from the lab bench. Therefore, it is common for technicians to stop work to manually record data on a paper form, then enter this into the MES or LIMS after the sample preparation is finished. This wastes time, means the digital copy is not the true original record, and increases the chance of documentation errors and protocol deviations.
Data integrity is a key component of GMP regulations and a major focus for regulatory agencies. Many pharmaceutical companies aim to follow the ALCOA+ principles of data integrity, where all data is attributable, legible, contemporaneous, original and accurate. The ALCOA+ principles are difficult to follow with paper or hybrid documentation systems.
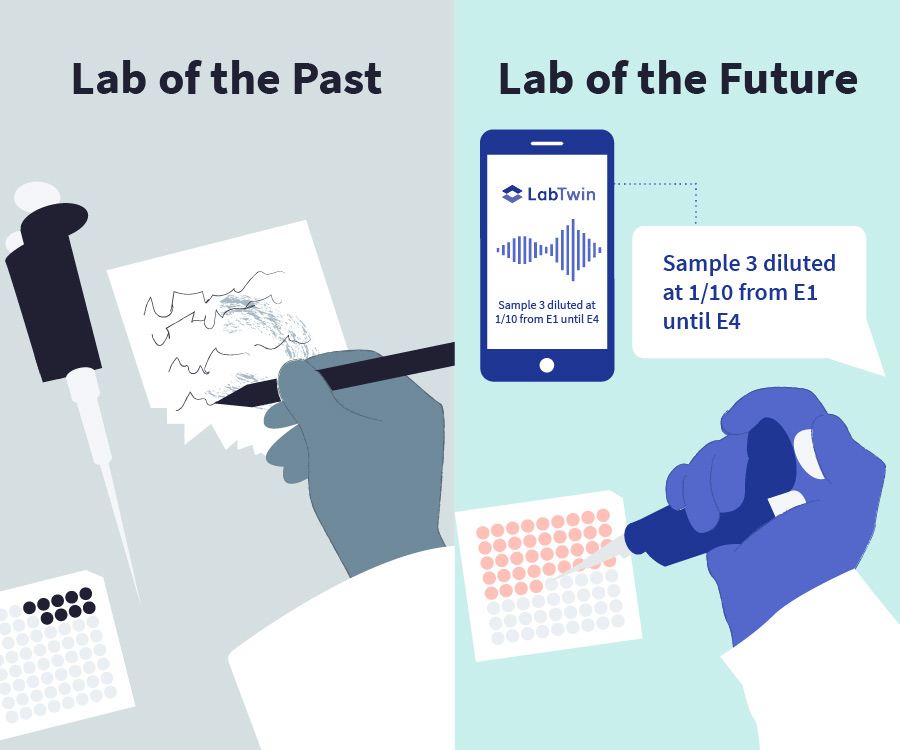
Voice-activated digital lab assistants help labs achieve all ALCOA+ principles and improve data integrity during GMP sample preparation. Digital lab assistants streamline workflows and facilitate data collection by:
- Reading out protocols step-by-step to technicians, ensuring protocol adherence
- Automatically connecting with lab instruments and recording data such as sample weights
- Collecting metadata including the temperature and humidity of the lab environment
- Allowing technicians to take voice notes about sample identification, reagent lot numbers, experimental conditions and any protocol deviations
- Automatically digitizing data, in real time, at the point of experimentation, so it is truly original and contemporaneous
- Attributing data with user identification, electronic signatures, and time and date stamps
- Storing data in a central, secure place with data encryption, private networks and user access control
These features eliminate distractions, save time, lower error rates and make GMP sample preparation more efficient.
Learn more about how voice-activated digital lab assistants help with GMP sample preparation in this white paper.










